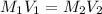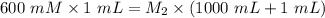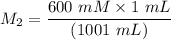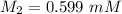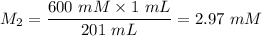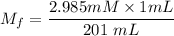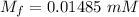Chemistry, 25.01.2021 20:50 kawaiiblurainbow
A purified protein is in a HEPES buffer at pH 7.0 with 600 mM NaCl. A 1 mL sample of the protein solution is dialyzed against 1.0 L of the same HEPES buffer with 0 mM NaCl. Once the dialysis has come to equilibrium, what is the concentration (in mM) of NaCl in the protein sample

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
A purified protein is in a HEPES buffer at pH 7.0 with 600 mM NaCl. A 1 mL sample of the protein sol...
Questions


English, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40


Business, 30.03.2021 03:40

Geography, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40



Mathematics, 30.03.2021 03:40

Mathematics, 30.03.2021 03:40

History, 30.03.2021 03:40

History, 30.03.2021 03:40




Mathematics, 30.03.2021 03:40