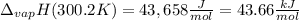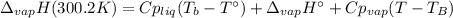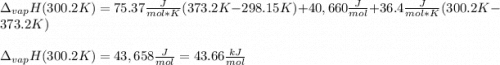Chemistry, 25.01.2021 20:40 PastelHibiscus
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar heat capacity of liquid water is 75.37 J K-1 mol-1 and that of gaseous water is 36.4 J K-1 mol-1. Assume that these values are independent of temperature. What is the heat of vaporization of water at 300.2 K?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Achef makes salad dressing by mixing oil, vinegar, and spices, as shown. which type of matter is the salad dressing?
Answers: 1

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1
You know the right answer?
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar h...
Questions

Mathematics, 26.09.2021 09:30

Biology, 26.09.2021 09:30



History, 26.09.2021 09:30



English, 26.09.2021 09:30

World Languages, 26.09.2021 09:30

English, 26.09.2021 09:30


History, 26.09.2021 09:30

Mathematics, 26.09.2021 09:30

Biology, 26.09.2021 09:30

Mathematics, 26.09.2021 09:30

Business, 26.09.2021 09:30

Mathematics, 26.09.2021 09:30

Chemistry, 26.09.2021 09:30

English, 26.09.2021 09:30