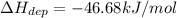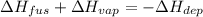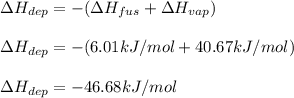Chemistry, 25.01.2021 14:00 nicoleh2015
The enthalpy of vaporization for water is 40.67 kJ/mol and the enthalpy of fusion for water is 6.01 kJ/mol. What is the enthalpy of deposition?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
The enthalpy of vaporization for water is 40.67 kJ/mol and the enthalpy of fusion for water is
6.01...
Questions

Chemistry, 13.03.2021 04:50

Mathematics, 13.03.2021 04:50

Spanish, 13.03.2021 04:50

Mathematics, 13.03.2021 04:50


Mathematics, 13.03.2021 04:50

English, 13.03.2021 04:50





Geography, 13.03.2021 04:50

History, 13.03.2021 04:50



Mathematics, 13.03.2021 04:50



English, 13.03.2021 04:50