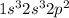Suppose you take a trip to a distant
you find that the periodic table there is derived from a...

Chemistry, 17.10.2019 19:20 jdisalle2808
Suppose you take a trip to a distant
you find that the periodic table there is derived from an arrangement of quantum numbers different from the one on earth. the rules in that universe are:
1. principal quantum number n = 1, 2, . . (as on earth);
2. angular momentum quantum number ℓ = 0, 1, 2,. . , n – 1 (as on earth);
3. magnetic quantum number mℓ = 0, 1, 2, . . , ℓ (only positive integers up to and including ℓ are allowed);
4. spin quantum number ms = –1, 0, 1 (that is, three allowed values of spin).
(a) assuming that the pauli exclusion principle remains valid, what is the maximum number of electrons that can populate a given orbital?
(b) write the electronic configuration of the element with atomic number 8 in the periodic table. (formatting: superscript numbers where appropriate but omit parentheses.)
(c) what is the atomic number of the second noble gas?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1



Chemistry, 23.06.2019 02:00
Which statement is true about the model of the electromagnetic spectrum a: it change the frequencies of light. b: it compare wavelengths of light. c: the color of light waves can be changed using the model. d: the intensities of light waves can be decreased using the model.
Answers: 2
You know the right answer?
Questions


Mathematics, 12.04.2021 06:00




Mathematics, 12.04.2021 06:00



Mathematics, 12.04.2021 06:00

English, 12.04.2021 06:00

Mathematics, 12.04.2021 06:00

Health, 12.04.2021 06:00

Mathematics, 12.04.2021 06:00




Mathematics, 12.04.2021 06:00