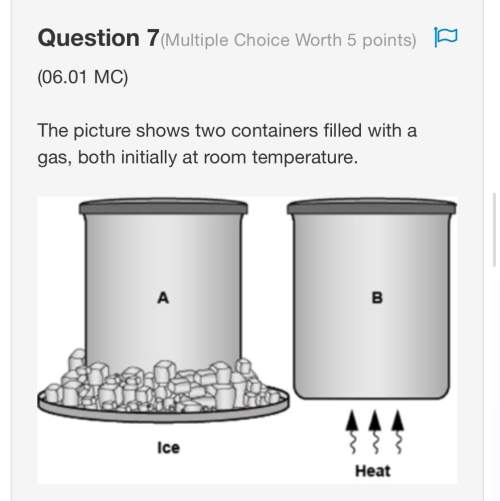
How will the temperatures of the water in the containers compare if an equal amount of heat is absorbed by each container of water without boiling?
A. Both water temperatures will increase, but container B's will increase more.
B. Both water temperatures will decrease, but container B's will decrease more.
C. Both water temperatures will increase by the same amount.
D. Both water temperatures will increase, but container A's will increase more.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Based on the law of conservation of energy, which statement is false? answer- energy is lost when machines dont work right
Answers: 1

Chemistry, 21.06.2019 20:40
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b.zinc7.14,c.copper 8.92,d.lead 11.34
Answers: 2


You know the right answer?
How will the temperatures of the water in the containers compare if an equal amount of heat is absor...
Questions

English, 14.01.2020 21:31

Computers and Technology, 14.01.2020 21:31


History, 14.01.2020 21:31

Mathematics, 14.01.2020 21:31


World Languages, 14.01.2020 21:31

English, 14.01.2020 21:31

Mathematics, 14.01.2020 21:31



Health, 14.01.2020 21:31

Mathematics, 14.01.2020 21:31




Chemistry, 14.01.2020 21:31

Mathematics, 14.01.2020 21:31

Mathematics, 14.01.2020 21:31