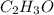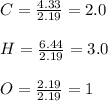Chemistry, 22.01.2021 19:00 jeffffffff
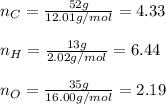
A percent composition analysis yields 52% carbon (C), 13% hydrogen (H), and 35% oxygen (O). What is the empirical formula for the compound?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
A percent composition analysis yields 52% carbon (C), 13% hydrogen (H), and 35% oxygen (O). What is...
Questions


Chemistry, 30.01.2020 07:59

History, 30.01.2020 07:59


Mathematics, 30.01.2020 07:59

Mathematics, 30.01.2020 07:59

Biology, 30.01.2020 07:59



Mathematics, 30.01.2020 07:59

Arts, 30.01.2020 07:59





Spanish, 30.01.2020 07:59

Mathematics, 30.01.2020 07:59