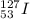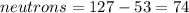Chemistry, 21.01.2021 22:50 kitttimothy55
Write the nuclide symbol for Iodine-127. How many protons, electrons, and neutrons are in this isotope of iodine

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
You know the right answer?
Write the nuclide symbol for Iodine-127. How many protons, electrons, and neutrons are in this isoto...
Questions




Mathematics, 05.05.2020 22:35

English, 05.05.2020 22:35


English, 05.05.2020 22:35

Mathematics, 05.05.2020 22:35

Mathematics, 05.05.2020 22:35



History, 05.05.2020 22:35

Biology, 05.05.2020 22:35

Mathematics, 05.05.2020 22:35

Mathematics, 05.05.2020 22:35

Engineering, 05.05.2020 22:35

Biology, 05.05.2020 22:35

Mathematics, 05.05.2020 22:35