
Chemistry, 21.01.2021 22:20 Jennifer312332
A 0.380 g sample of an unknown monoprotic acid is titrated with 0.300 M KOH to the equivalence point. It requires 20.1 mL of KOH to reach this point. Elemental analysis is also performed, showing that the acid contains 1.6% H, 22.2% N, and 76.2% O. Use this information to determine the molecular formula and the molar mass of the unknown acid.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2


Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1
You know the right answer?
A 0.380 g sample of an unknown monoprotic acid is titrated with 0.300 M KOH to the equivalence point...
Questions


History, 15.02.2021 06:40


Social Studies, 15.02.2021 06:40



Mathematics, 15.02.2021 06:40



Mathematics, 15.02.2021 06:40


Mathematics, 15.02.2021 06:40




Business, 15.02.2021 06:40

Spanish, 15.02.2021 06:40



English, 15.02.2021 06:40

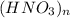 ( let )
( let ) 

