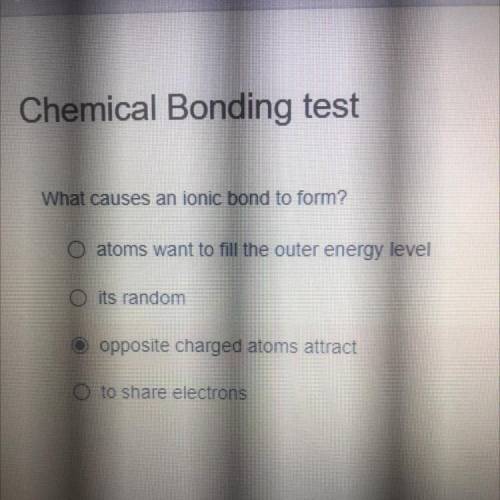
What causes an ionic bond to form?
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2


Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
You know the right answer?
Questions


Mathematics, 06.05.2020 04:08


Mathematics, 06.05.2020 04:08

Mathematics, 06.05.2020 04:08

Mathematics, 06.05.2020 04:08





Physics, 06.05.2020 04:08


English, 06.05.2020 04:08

Mathematics, 06.05.2020 04:08

English, 06.05.2020 04:08

History, 06.05.2020 04:08

Mathematics, 06.05.2020 04:08

Mathematics, 06.05.2020 04:08