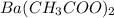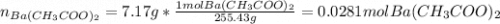Chemistry, 19.01.2021 19:50 bercishicicorbin
Suppose 7.17g of barium acetate is dissolved in 350mL of a 79.0mM aqueous solution of sodium chromate. Calculate the final molarity of barium cation in the solution. You can assume the volume of the solution doesn't change when the barium acetate is dissolved in it. Round your answer to 1 significant digit.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Suppose 7.17g of barium acetate is dissolved in 350mL of a 79.0mM aqueous solution of sodium chromat...
Questions





Mathematics, 23.03.2021 04:40


Business, 23.03.2021 04:40


Mathematics, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40

History, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40



History, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40

Mathematics, 23.03.2021 04:40

Social Studies, 23.03.2021 04:40