
Chemistry, 19.01.2021 01:00 zarakanchi
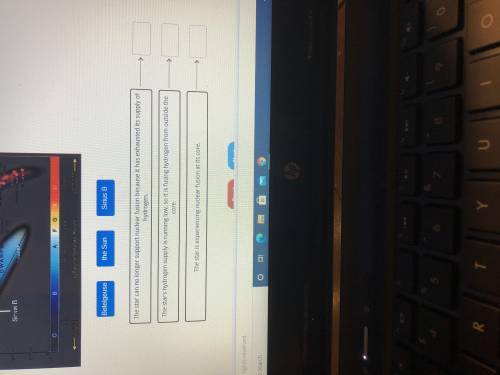
Using the hertz sprung Russell diagram to determine which condition describes each door use the arrows to help locate the stars

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 21.06.2019 22:30
Write the symbol for every chemical element that has atomic number greater than 3 and atomic mass less than 12.0 u.
Answers: 1

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2
You know the right answer?
Using the hertz sprung Russell diagram to determine which condition describes each door use the arro...
Questions


Business, 03.07.2019 21:30


Social Studies, 03.07.2019 21:30

Social Studies, 03.07.2019 21:30




Mathematics, 03.07.2019 21:30

Social Studies, 03.07.2019 21:30

Mathematics, 03.07.2019 21:30

Mathematics, 03.07.2019 21:30


Mathematics, 03.07.2019 21:30

Biology, 03.07.2019 21:30

English, 03.07.2019 21:30



History, 03.07.2019 21:30

History, 03.07.2019 21:30



