
Hydrobromic acid dissolves solid iron according to the following reaction:
Fe(s)+2HBr(aq)→FeBr2(aq)+H2(g)
What mass of HBr (in g) would you need to dissolve a 2.8-g pure iron bar on a padlock?
What mass of H2 would be produced by the complete reaction of the iron bar?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1

Chemistry, 23.06.2019 01:30
Concentrations expressed as a percent by mass are useful when the solute is a a. liquid b. gas c. solid
Answers: 1

Chemistry, 23.06.2019 08:00
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3
You know the right answer?
Hydrobromic acid dissolves solid iron according to the following reaction:
Fe(s)+2HBr(aq)→FeBr2(aq...
Questions


Mathematics, 12.12.2019 01:31


Health, 12.12.2019 01:31




History, 12.12.2019 01:31

English, 12.12.2019 01:31



Mathematics, 12.12.2019 01:31


Mathematics, 12.12.2019 01:31

Physics, 12.12.2019 01:31

History, 12.12.2019 01:31

Geography, 12.12.2019 01:31


Mathematics, 12.12.2019 01:31

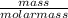
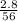 = 0.05mole
= 0.05mole 

