
Chemistry, 14.01.2021 14:00 darkcrystal463
0.2 mol of hydrocarbons undergo complete combustion to give 35.2 of carbon dioxide and 14.4g of water as the only product. What is the molecular formula of hydrocarbon

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3

Chemistry, 23.06.2019 05:00
Which characteristics affect ocean water’s temperature? check all that apply. depth location mass salinity waves
Answers: 1

Chemistry, 23.06.2019 07:30
Assume that 13.5 g solid aluminum (al) react with hcl to produce solid aluminum chloride (alcl3) salt and gaseous hydrogen (h2) at standard temperature and pressure.
Answers: 1
You know the right answer?
0.2 mol of hydrocarbons undergo complete combustion to give 35.2 of carbon dioxide and 14.4g of wate...
Questions

Mathematics, 24.04.2020 20:16

Mathematics, 24.04.2020 20:16

Mathematics, 24.04.2020 20:16


Mathematics, 24.04.2020 20:17

Chemistry, 24.04.2020 20:17

Spanish, 24.04.2020 20:17



Social Studies, 24.04.2020 20:17

Computers and Technology, 24.04.2020 20:17





English, 24.04.2020 20:17


English, 24.04.2020 20:17

Mathematics, 24.04.2020 20:17

Biology, 24.04.2020 20:17

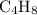 .
.  ,
, 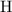 , and
, and  on a modern periodic table:
on a modern periodic table: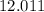 .
.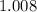 .
.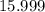 .
.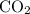 and
and 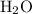 :
: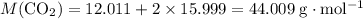 ;
;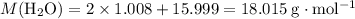 .
.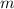 of
of 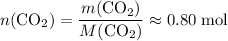 ;
;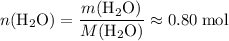 .
.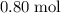 of
of 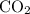 contains
contains 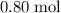 of
of  atoms.Each
atoms.Each 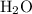 contains
contains 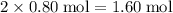 of
of 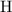 atoms.
atoms.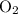 .
.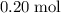 of this unknown hydrocarbon molecules contains:
of this unknown hydrocarbon molecules contains: 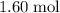 of
of 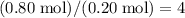 carbon atoms and
carbon atoms and 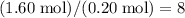 hydrogen atoms.
hydrogen atoms.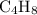 .
.

