

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 23.06.2019 08:00
How many distinct monochlorinated products, including stereoisomers, can result when the alkane below is heated in the presence of cl2? 3 4 5 6 7?
Answers: 3
You know the right answer?
Read the chemical equation. Fe2O3 + CO Fe + CO2 If 2 moles of Fe2O3 react with 9 moles of Co, how ma...
Questions

Biology, 04.12.2019 14:31



Social Studies, 04.12.2019 14:31

History, 04.12.2019 14:31

Social Studies, 04.12.2019 14:31


Social Studies, 04.12.2019 14:31

History, 04.12.2019 14:31



Physics, 04.12.2019 14:31





Mathematics, 04.12.2019 14:31


Social Studies, 04.12.2019 14:31

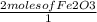 x
x 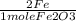 = 4 Fe
= 4 Fe 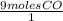 x
x 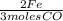 = 6 moles CO2
= 6 moles CO2


