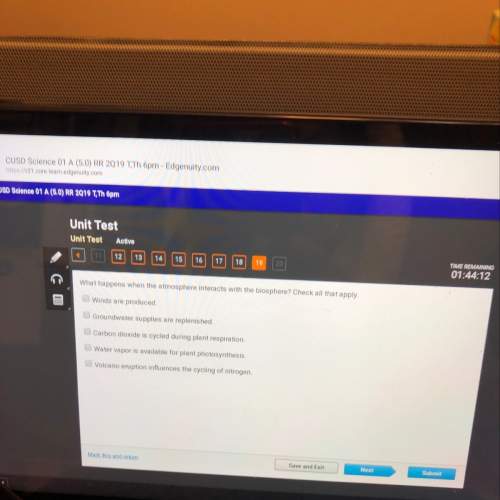
Chemistry, 13.01.2021 18:20 1930isainunez
PLS HELP ASAP
In the process of creating ammonia (NH3), nitrogen gas (N2 ) is combined with hydrogen gas (H2) that is extracted from hydrocarbons such as methane (CH4) or propane (C3H8) gas. Manufacturers must determine the appropriate amount of each reactant so as to produce the highest yield of product.
The balanced reaction for ammonia is below:
N2+2H3→2NH3
Explain why determining the limiting reactant is important to a manufacturer trying to produce NH3 and the effect of running out of reactant.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2


Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1
You know the right answer?
PLS HELP ASAP
In the process of creating ammonia (NH3), nitrogen gas (N2 ) is combined with hydroge...
Questions

Social Studies, 22.07.2021 14:00

History, 22.07.2021 14:00

English, 22.07.2021 14:00


Social Studies, 22.07.2021 14:00


Mathematics, 22.07.2021 14:00



Mathematics, 22.07.2021 14:00

Mathematics, 22.07.2021 14:00

English, 22.07.2021 14:00

History, 22.07.2021 14:00

Chemistry, 22.07.2021 14:00


Physics, 22.07.2021 14:00


Mathematics, 22.07.2021 14:00

Chemistry, 22.07.2021 14:00