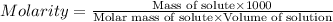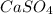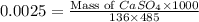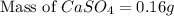Chemistry, 11.01.2021 17:50 Perdikaris
A chemist adds 485 mL of a 0.0025 mol/L calcium sulfate solution to a reaction flask. Calculate the mass in grams of calcium sulfate the chemist has added to the flask. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 23.06.2019 01:00
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1
You know the right answer?
A chemist adds 485 mL of a 0.0025 mol/L calcium sulfate solution to a reaction flask. Calculate the...
Questions


English, 11.03.2020 09:35

Chemistry, 11.03.2020 09:38

Mathematics, 11.03.2020 09:38


Biology, 11.03.2020 09:38


English, 11.03.2020 09:39


Chemistry, 11.03.2020 09:39

History, 11.03.2020 09:39



English, 11.03.2020 09:41

Mathematics, 11.03.2020 09:42



Chemistry, 11.03.2020 09:47


Mathematics, 11.03.2020 09:49