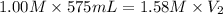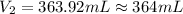Chemistry, 11.01.2021 16:10 makailaaa2
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do this by pouring out some 1.58M aqueous sodium carbonate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in mL of the sodium carbonate stock solution that the chemist should pour out. Round your answer to 3 significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 16:00
How does blood clotting prevent the entry of pathogens through cuts and wounds? answer asap,, this is due tomorrow. will mark as brainliest or whatever you call it : )
Answers: 2

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do th...
Questions

Mathematics, 08.10.2019 07:20


Computers and Technology, 08.10.2019 07:20

History, 08.10.2019 07:20

Biology, 08.10.2019 07:20

English, 08.10.2019 07:20

Biology, 08.10.2019 07:20



Mathematics, 08.10.2019 07:20


Physics, 08.10.2019 07:20


Social Studies, 08.10.2019 07:20




Social Studies, 08.10.2019 07:20


Computers and Technology, 08.10.2019 07:20

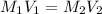
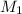 = molarity of aqueous sodium carbonate
= molarity of aqueous sodium carbonate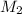 = molarity of aqueous sodium carbonate stock solution
= molarity of aqueous sodium carbonate stock solution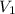 = volume of aqueous sodium carbonate
= volume of aqueous sodium carbonate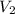 = volume of aqueous sodium carbonate stock solution
= volume of aqueous sodium carbonate stock solution