
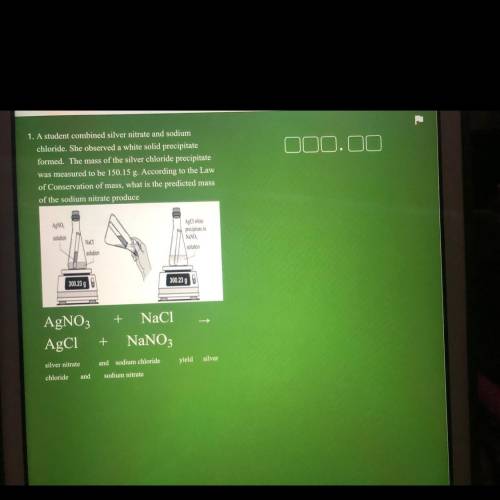
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
formed. The mass of the silver chloride precipitate
was measured to be 150.15 g. According to the Law
of Conservation of mass, what is the predicted mass
of the sodium nitrate produce
AgNO
AgCl white
precipitate in
solution
Naci
NaNO
solution
solution
300.23 g
300.23 90
+ NaCl
AgNO3
AgC1
+
NaNO3
silver nitrate
and sodium chloride
yield silver
chloride
and
sodium nitrate

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which of these sequences lists the correct order for the creation of sedimentary rock from sediment? a. deposition, burial, compaction, cementation b. burial, deposition, compaction, cementation c. compaction, deposition, burial, cementation d. cementation, deposition, burial, compaction
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1
You know the right answer?
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
Questions

Mathematics, 04.02.2020 10:44








Social Studies, 04.02.2020 10:44

Mathematics, 04.02.2020 10:44

History, 04.02.2020 10:44


History, 04.02.2020 10:44

English, 04.02.2020 10:44


Computers and Technology, 04.02.2020 10:44

Mathematics, 04.02.2020 10:44

English, 04.02.2020 10:44

Mathematics, 04.02.2020 10:45

English, 04.02.2020 10:45



