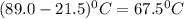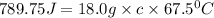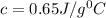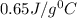Chemistry, 06.01.2021 20:20 koryhudson8124
An 18.0 g piece of an unidentified metal was heated from 21.5 °C to 89.0 °C. If 789.75 J of heat energy was absorbed by the metal in the heating process, what was the identity of the metal?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
An 18.0 g piece of an unidentified metal was heated from 21.5 °C to 89.0 °C. If 789.75 J of heat ene...
Questions

Mathematics, 10.03.2021 22:20


Chemistry, 10.03.2021 22:20


English, 10.03.2021 22:20

Chemistry, 10.03.2021 22:20


Social Studies, 10.03.2021 22:20

English, 10.03.2021 22:20

Mathematics, 10.03.2021 22:20


Mathematics, 10.03.2021 22:20


Mathematics, 10.03.2021 22:20

Mathematics, 10.03.2021 22:20

World Languages, 10.03.2021 22:20

Biology, 10.03.2021 22:20

Mathematics, 10.03.2021 22:20


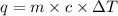
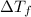 = final temperature - initial temperature =
= final temperature - initial temperature =