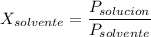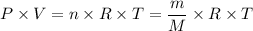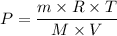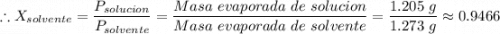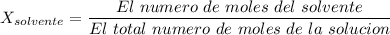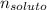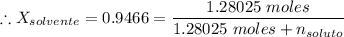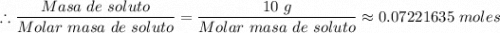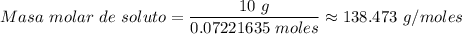Chemistry, 05.01.2021 16:50 sbanoali6120
Si se encierran 100 gramos de benceno C6H6 en un recipiente con aire, se observa que, al cabo de cierto tiempo, se han evaporado 1,273 gramos, logr´andose as´ı la saturaci´on del aire con vapor de benceno. En cambio, si se disuelven 10 gramos de un soluto no vol´atil en 100 gramos de benceno y la mezcla se coloca en el recipiente anterior y a la misma temperatura, se observa que se han evaporado 1,205 gramos de benceno. A partir de estos datos, deducir la masa molecular de la sustancia disuelta.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1


Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Si se encierran 100 gramos de benceno C6H6 en un recipiente con aire, se observa que, al cabo de cie...
Questions







History, 06.05.2020 22:58

Mathematics, 06.05.2020 22:58

Mathematics, 06.05.2020 22:58

Mathematics, 06.05.2020 22:58



English, 06.05.2020 22:58



Chemistry, 06.05.2020 22:58


Biology, 06.05.2020 22:58


English, 06.05.2020 22:58

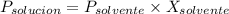
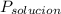 = La presión de vapor de la solución.
= La presión de vapor de la solución.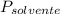 = La presión de vapor del solvente
= La presión de vapor del solvente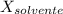 = La fracción molar del solvente
= La fracción molar del solvente