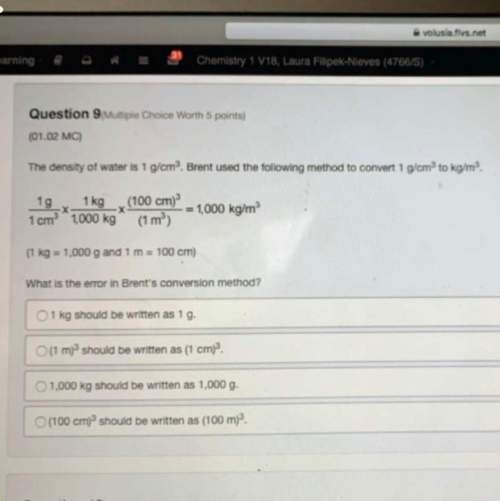
NH4+ + NO2– ⟶ N2 + 2H2O

Chemistry, 02.01.2021 04:10 luludawn3874
Two answer multiple choice-- please help!
Study the reaction.
NH4+ + NO2– ⟶ N2 + 2H2O
The rate of this reaction is proportional to the concentration of ammonium (NH4+) and the concentration of nitrogen dioxide (NO2–).
What is the rate law of this reaction?
Rate = K[NH4+][NO2–]
Rate = K[N2][H2O] ^2

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
Two answer multiple choice-- please help!
Study the reaction.
NH4+ + NO2– ⟶ N2 + 2H2O
NH4+ + NO2– ⟶ N2 + 2H2O
Questions


Mathematics, 20.08.2021 22:30



English, 20.08.2021 22:30

Mathematics, 20.08.2021 22:30


Biology, 20.08.2021 22:30

Mathematics, 20.08.2021 22:30

Biology, 20.08.2021 22:30

Advanced Placement (AP), 20.08.2021 22:30

Mathematics, 20.08.2021 22:30


Biology, 20.08.2021 22:30

Mathematics, 20.08.2021 22:30

Mathematics, 20.08.2021 22:30