
Chemistry, 02.01.2021 02:10 cdradlet2001
SO2Cl2 rightarrow SO2 (g) + Cl2 (g)At 600 K, SO2Cl2 will decompose to form sulfure dioxide and chlorine gas via the above equation. If the reaction is found to be first order overall, which of the following will cause an increase in he half life of SO2CL2?A. Increasing the initial concentration of SO2,CI2.B. Increasing the temperature at which the reaction occurs. C. Decreasing the overall pressure in the container. D. None of these will increase the half life.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2



Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2
You know the right answer?
SO2Cl2 rightarrow SO2 (g) + Cl2 (g)At 600 K, SO2Cl2 will decompose to form sulfure dioxide and chlor...
Questions

Physics, 30.06.2021 21:10

Mathematics, 30.06.2021 21:10

Mathematics, 30.06.2021 21:10

History, 30.06.2021 21:10

English, 30.06.2021 21:10

Computers and Technology, 30.06.2021 21:10





English, 30.06.2021 21:10

Mathematics, 30.06.2021 21:10

Social Studies, 30.06.2021 21:10

Mathematics, 30.06.2021 21:10


English, 30.06.2021 21:10




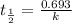
 following first order kinetics, none of the factors will increase the half life.
following first order kinetics, none of the factors will increase the half life.

