

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

You know the right answer?
List six elements with only 1 electron in an s orbital...
Questions

Mathematics, 05.05.2021 05:40

Mathematics, 05.05.2021 05:40

English, 05.05.2021 05:40




Social Studies, 05.05.2021 05:40

History, 05.05.2021 05:40



English, 05.05.2021 05:40

English, 05.05.2021 05:40


Mathematics, 05.05.2021 05:40

Mathematics, 05.05.2021 05:40

Mathematics, 05.05.2021 05:40


Mathematics, 05.05.2021 05:40

History, 05.05.2021 05:40

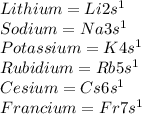
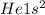
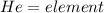
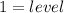
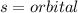
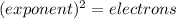
 , representative of a single electron in their s-orbital.
, representative of a single electron in their s-orbital. 

