
Chemistry, 16.12.2020 22:50 ceceshelby1130
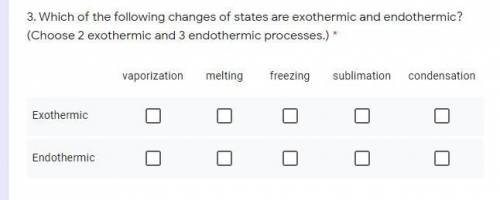
Which of the following changes of states are exothermic and endothermic? (Choose 2 exothermic and 3 endothermic processes.)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:50
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
Which of the following changes of states are exothermic and endothermic? (Choose 2 exothermic and 3...
Questions


History, 12.11.2019 20:31

Social Studies, 12.11.2019 20:31



Biology, 12.11.2019 20:31

Mathematics, 12.11.2019 20:31

History, 12.11.2019 20:31

English, 12.11.2019 20:31


Mathematics, 12.11.2019 20:31

Biology, 12.11.2019 20:31

Biology, 12.11.2019 20:31

English, 12.11.2019 20:31


Mathematics, 12.11.2019 20:31


Mathematics, 12.11.2019 20:31

Chemistry, 12.11.2019 20:31

Mathematics, 12.11.2019 20:31



