
Chemistry, 16.12.2020 21:30 zoeyjorschumb17
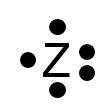
Would this atom gain or lose electrons and what would be the resulting charge?
A) Lose 3 electrons, 3+ charge
B) Lose 3 electrons, 3- charge
C) Gain 3 electrons, 3- charge
D) Gain 3 electrons, 3+ charge

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2



Chemistry, 23.06.2019 13:30
32p and 31p are two isotopes of phosphorus. compare the number if subatomic particles that are present in the atoms of these isotopes.
Answers: 1
You know the right answer?
Would this atom gain or lose electrons and what would be the resulting charge?
A) Lose 3 electrons,...
Questions


Advanced Placement (AP), 12.02.2021 16:10





Chemistry, 12.02.2021 16:10




History, 12.02.2021 16:10







Mathematics, 12.02.2021 16:10


Mathematics, 12.02.2021 16:10



