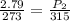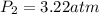Chemistry, 15.12.2020 15:50 Rainey1664
What is the resultant pressure if 1.7 mol of ideal gas at 273 K and 2.79 atm in a closed container of constant volume is heated to 315 K? Answer in units of atm.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2
You know the right answer?
What is the resultant pressure if 1.7 mol of ideal gas at 273 K and 2.79 atm in a closed container o...
Questions




Mathematics, 25.07.2019 18:00





Business, 25.07.2019 18:00


Social Studies, 25.07.2019 18:00

Biology, 25.07.2019 18:00




Social Studies, 25.07.2019 18:00

English, 25.07.2019 18:00


Mathematics, 25.07.2019 18:00

Biology, 25.07.2019 18:00

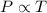 (At constant volume and number of moles)
(At constant volume and number of moles)
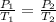
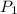 = initial pressure of gas = 2.79 atm
= initial pressure of gas = 2.79 atm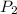 = final pressure of gas = ?
= final pressure of gas = ?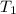 = initial temperature of gas = 273K
= initial temperature of gas = 273K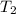 = final temperature of gas = 315 K
= final temperature of gas = 315 K