Chemistry, 15.12.2020 01:00 jamieric0324
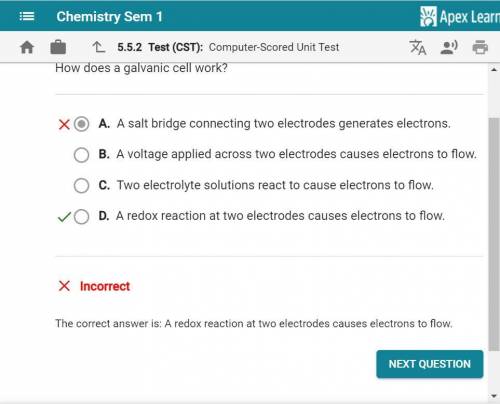
How does a galvanic cell work? A. A voltage applied across two electrodes causes electrons to flow. B. A salt bridge connecting two electrodes generates electrons. O C. A redox reaction at two electrodes causes electrons to flow. D. Two electrolyte solutions react to cause electrons to flow.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1



Chemistry, 23.06.2019 04:31
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1
You know the right answer?
How does a galvanic cell work? A. A voltage applied across two electrodes causes electrons to flow....
Questions



History, 20.10.2020 04:01

English, 20.10.2020 04:01







Mathematics, 20.10.2020 04:01


English, 20.10.2020 04:01

Social Studies, 20.10.2020 04:01

History, 20.10.2020 04:01


Mathematics, 20.10.2020 04:01



Mathematics, 20.10.2020 04:01