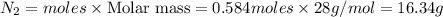What mass of N2 will be needed to produce 31.5 grams of N2O5?
4N2 + 502 --> 2N2O5
a) 158.3...


Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2


Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
Questions

Mathematics, 25.10.2019 01:43




Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43

English, 25.10.2019 01:43

Social Studies, 25.10.2019 01:43



Biology, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43


Mathematics, 25.10.2019 01:43


Mathematics, 25.10.2019 01:43

Mathematics, 25.10.2019 01:43


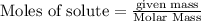
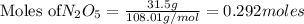
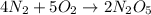
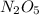 are produced by= 4 moles of
are produced by= 4 moles of 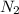
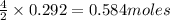 of
of