

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 19:30
Which one of the following substances would be the most soluble in ccl4? na2so4 h2o ch3ch2ch2ch2oh c4h10 hi
Answers: 1

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
A 2.89 g sample of a metal chloride, MCl2, is dissolved in water and treated with excess aqueous sil...
Questions

Mathematics, 31.05.2021 19:30

Mathematics, 31.05.2021 19:30

Social Studies, 31.05.2021 19:30






Mathematics, 31.05.2021 19:30





Mathematics, 31.05.2021 19:30


Mathematics, 31.05.2021 19:30


Mathematics, 31.05.2021 19:30

World Languages, 31.05.2021 19:30

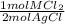 = 0.0224 mol MCl₂
= 0.0224 mol MCl₂


