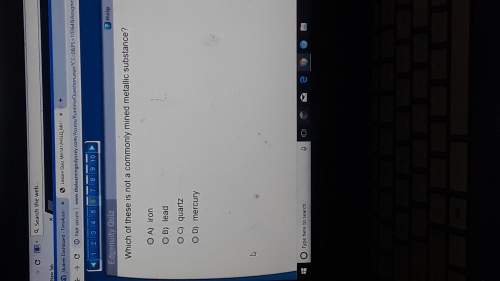
Chemistry, 12.12.2020 16:30 4presidents
Why is density a property but mass and volume is not a property?

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2
You know the right answer?
Why is density a property but mass and volume is not a property?...
Questions

Biology, 12.10.2019 16:50





Chemistry, 12.10.2019 16:50



Mathematics, 12.10.2019 17:00

Geography, 12.10.2019 17:00







Mathematics, 12.10.2019 17:00


Chemistry, 12.10.2019 17:00

Mathematics, 12.10.2019 17:00