
Chemistry, 10.12.2020 20:50 YaBoiMando2061
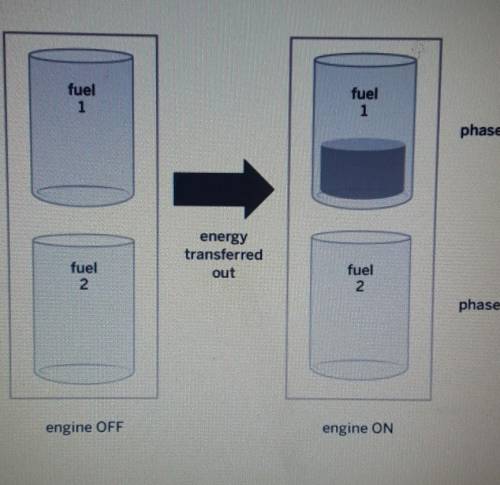
PLEASE ANSWER A certain type of ship has two tanks in its engine. Each tank contains a different type of fuel. When the engine turns on, the same amount of energy is transferred out of both fuels as shown in the diagram below. Why did fuell change phase. but fuel 2 stayed the same? Explain what happened to the molecules of both fuels.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1

Chemistry, 23.06.2019 05:00
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius.a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit.a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius.a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1
You know the right answer?
PLEASE ANSWER
A certain type of ship has two tanks in its engine. Each tank contains a different ty...
Questions

Health, 05.02.2021 01:00


Medicine, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00





Health, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00


Mathematics, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00

Biology, 05.02.2021 01:00

History, 05.02.2021 01:00




Mathematics, 05.02.2021 01:00



