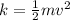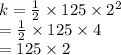Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 23.06.2019 04:00
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1

Chemistry, 23.06.2019 06:00
Give one example of a pure (exact) number and of an estimated (measured) number.
Answers: 2

Chemistry, 23.06.2019 09:00
What sources of error may have contributed to the percent yield not being 100 percent? think about things that may have led to inaccurate measurements or where mass of the product could have been lost if this experiment was conducted in a physical laboratory.
Answers: 2
You know the right answer?
What is the kinetic energy of a 125 kg cart that is moving at a speed of 2 m/s?
(i suck at science...
Questions


Chemistry, 15.12.2020 18:10



Mathematics, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10


Physics, 15.12.2020 18:10


Mathematics, 15.12.2020 18:10




Mathematics, 15.12.2020 18:10


Mathematics, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10


History, 15.12.2020 18:10