
Chemistry, 10.12.2020 04:00 lizzyhearts
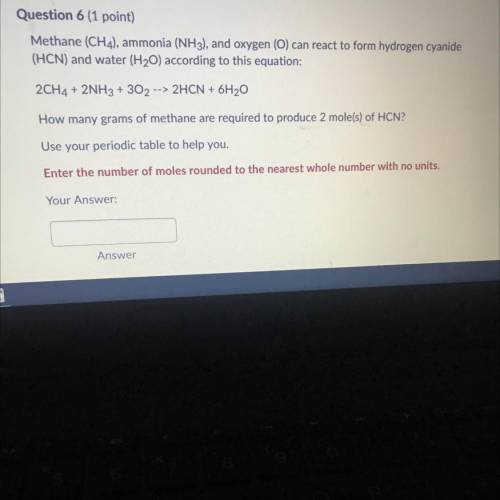
Methane (CH 4 ) , ammonia (NH 3 ) , and oxygen () can reaci to form hydrogen cyanide (HCN) and water (H 2 O) according to this equation 2CH 4 +2NH 3 +3O 2 2HCN+6H 2 O How many grams of methane are required to produce 2 moles ? Use your periodic table to help you. Please show work

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1

You know the right answer?
Methane (CH 4 ) , ammonia (NH 3 ) , and oxygen () can reaci to form hydrogen cyanide (HCN) and water...
Questions

Chemistry, 10.11.2020 20:00

Chemistry, 10.11.2020 20:00




World Languages, 10.11.2020 20:00

Computers and Technology, 10.11.2020 20:00



Mathematics, 10.11.2020 20:00


Mathematics, 10.11.2020 20:00

Mathematics, 10.11.2020 20:00

Mathematics, 10.11.2020 20:00

Mathematics, 10.11.2020 20:00

Social Studies, 10.11.2020 20:00

Mathematics, 10.11.2020 20:00

Mathematics, 10.11.2020 20:00


Mathematics, 10.11.2020 20:00



