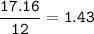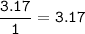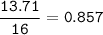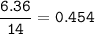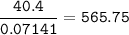Chemistry, 06.12.2020 05:00 timithythaxton
A 40.4 g sample of a protein contains 17.16 g of carbon, 3.17 g of hydrogen, 13.71 g of oxygen, and the rest being nitrogen. This 40.4-gram sample is known to be 0.07141 moles. Determine the molecular formula of this protein.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1


Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
A 40.4 g sample of a protein contains 17.16 g of carbon, 3.17 g of hydrogen, 13.71 g of oxygen, and...
Questions

Biology, 06.07.2019 13:20


Biology, 06.07.2019 13:20




History, 06.07.2019 13:20

Chemistry, 06.07.2019 13:20


Biology, 06.07.2019 13:20


Business, 06.07.2019 13:20



Biology, 06.07.2019 13:20


History, 06.07.2019 13:20



History, 06.07.2019 13:20