PLEASE HELP ME PLEASE I NEED THE ANSWER QUICK
...

Chemistry, 04.12.2020 21:10 haileybland8050
PLEASE HELP ME PLEASE I NEED THE ANSWER QUICK
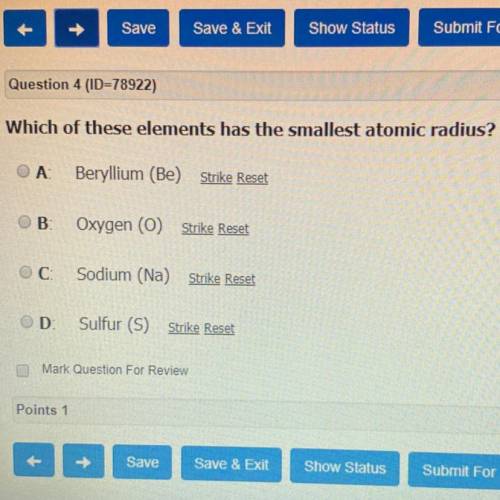

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2


Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Questions

Mathematics, 13.01.2020 03:31

Mathematics, 13.01.2020 03:31



Mathematics, 13.01.2020 03:31

English, 13.01.2020 03:31


History, 13.01.2020 03:31

Geography, 13.01.2020 03:31

English, 13.01.2020 03:31

Mathematics, 13.01.2020 03:31

History, 13.01.2020 03:31





Physics, 13.01.2020 03:31

History, 13.01.2020 03:31

Mathematics, 13.01.2020 03:31

French, 13.01.2020 03:31



