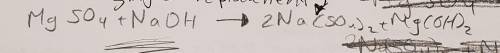Chemistry, 02.12.2020 21:30 irvinbhangal2
3. Magnesium sulfate is added to sodium hydroxide to produce sodium sulfate and magnesium hydroxide (10 points)
a. Write the skeletal equation and Balance the chemical equation describing the reaction above.
b. What kind of reaction is this?
c. Rewrite the balanced chemical equation, adding state symbols for all compounds in the reaction.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1
You know the right answer?
3. Magnesium sulfate is added to sodium hydroxide to produce sodium sulfate and magnesium hydroxide...
Questions


Business, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01



Advanced Placement (AP), 05.10.2020 14:01

History, 05.10.2020 14:01



English, 05.10.2020 14:01

Geography, 05.10.2020 14:01


English, 05.10.2020 14:01

Social Studies, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01

Health, 05.10.2020 14:01

Spanish, 05.10.2020 14:01