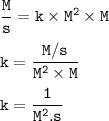4. For the reaction A + 2B → C, the rate law is
Δ[C]/ΔT = k[A]²[B]
What are the units of the...

Chemistry, 02.12.2020 19:20 MorrisAshley
4. For the reaction A + 2B → C, the rate law is
Δ[C]/ΔT = k[A]²[B]
What are the units of the rate constant where time is measured in seconds?
a. 1/ M · s
b. 1/ M² · s
c. 1/ s
d. M²/ s
e. M/ s

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1

Chemistry, 23.06.2019 01:00
The time that is taken by neptune once around the sun is called
Answers: 1

You know the right answer?
Questions

Mathematics, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30

Physics, 02.07.2019 09:30

History, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30

History, 02.07.2019 09:30

Computers and Technology, 02.07.2019 09:30

History, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30

Mathematics, 02.07.2019 09:30




History, 02.07.2019 09:30


Mathematics, 02.07.2019 09:30


History, 02.07.2019 09:30

![\large{\boxed{\boxed{\bold{v~=~k.[A]^a[B]^b}}}](/tpl/images/0942/2153/cb324.png)