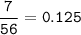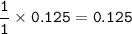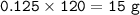Chemistry, 27.11.2020 14:00 whitakers87
Sulfur dioxide is an atmospheric pollutant.
Sulfur dioxide pollution is reduced by reacting calcium oxide with sulfur dioxide to
produce calcium sulfite.
CaO + SO2 → CaSO3
7.00 g of calcium oxide reacts with an excess of sulfur dioxide.
Relative atomic masses (Ar): O = 16 S = 32 Ca = 40 Calculate the mass of calcium sulfite produced


Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
Sulfur dioxide is an atmospheric pollutant.
Sulfur dioxide pollution is reduced by reacting calcium...
Questions

History, 24.08.2019 07:00

English, 24.08.2019 07:00


Mathematics, 24.08.2019 07:00


Social Studies, 24.08.2019 07:00

Mathematics, 24.08.2019 07:00


Biology, 24.08.2019 07:00

Biology, 24.08.2019 07:00


Biology, 24.08.2019 07:00

Mathematics, 24.08.2019 07:00



History, 24.08.2019 07:00

Physics, 24.08.2019 07:00

Chemistry, 24.08.2019 07:00