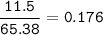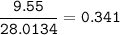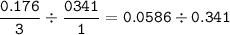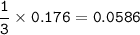Chemistry, 24.11.2020 19:10 natalia9573
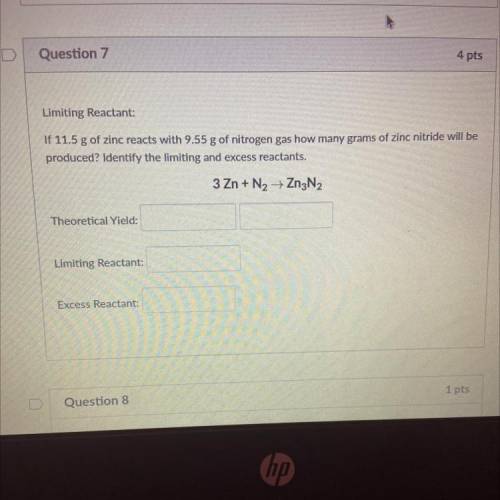
Limiting Reactant:
If 11.5 g of zinc reacts with 9.55 g of nitrogen gas how many grams of zinc nitride will be
produced? Identify the limiting and excess reactants.
3 Zn + N2 + Zn3N2
Theoretical Yield:
Limiting Reactant:
Excess Reactant:

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In an energy pyramid, which level has the most available energy?
Answers: 1

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1
You know the right answer?
Limiting Reactant:
If 11.5 g of zinc reacts with 9.55 g of nitrogen gas how many grams of zinc nitr...
Questions

History, 21.09.2019 16:10



History, 21.09.2019 16:10





Physics, 21.09.2019 16:10



Biology, 21.09.2019 16:10


Mathematics, 21.09.2019 16:10

History, 21.09.2019 16:10

Mathematics, 21.09.2019 16:10