
Chemistry, 23.11.2020 20:50 arangoaiden71
Calculate the change of enthalpy for the reaction CH4 (g) + NH3 (g) --> HCN (g) +3H2 (g) from the following reactions:
Reaction 1: N2 (g) + 3H2 (g) --> 2NH3 (g) Change in enthalpy: -91.8 kJ/mol
Reaction 2: C (s, graphite) + 2H2 (g) --> CH4 (g) Change in enthalply: -74.9 kJ/mol
Reaction 3: H2 (g) + 2C (s, graphite) + N2 (g) --> 2HCN (g) Change in enthalpy: +270.3 kJ/mol
Include the following:
The numerical answer with correct units.
State which reactions, if any, you had to "Flip".
State which reactions you had to multiply, if any, to get the correct amount of the compound.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 23.06.2019 09:30
Where are the noble gases located in the periodic table? a. in the center b. on the left side c. in the upper right corner d. on the far right side
Answers: 1

You know the right answer?
Calculate the change of enthalpy for the reaction CH4 (g) + NH3 (g) --> HCN (g) +3H2 (g) from the...
Questions


History, 02.01.2020 09:31


Spanish, 02.01.2020 09:31

Chemistry, 02.01.2020 09:31


Mathematics, 02.01.2020 09:31


English, 02.01.2020 09:31

Biology, 02.01.2020 09:31


Mathematics, 02.01.2020 09:31



Business, 02.01.2020 09:31

Mathematics, 02.01.2020 09:31


Mathematics, 02.01.2020 09:31


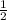 in order to have it cancel out with the other reactions. For the third reaction, we'd want to we'd want to switch the products/reactants and multiply the coefficients by
in order to have it cancel out with the other reactions. For the third reaction, we'd want to we'd want to switch the products/reactants and multiply the coefficients by 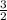 H₂ --> NH₃ (ΔH=-45.9)
H₂ --> NH₃ (ΔH=-45.9)

