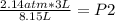Chemistry, 20.11.2020 14:00 falishaduncanovmtz2
A sample of gas has an initial volume of 3.00 L and an initial pressure of 2.14 atm. If the volume expands to 8.15 L, what is the final pressure?a. 0.855 atm b. 0.788 atm c. 3.49 atm d. 5.81 atm

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
A sample of gas has an initial volume of 3.00 L and an initial pressure of 2.14 atm. If the volume e...
Questions


History, 13.10.2019 12:30


Computers and Technology, 13.10.2019 12:30



History, 13.10.2019 12:30





History, 13.10.2019 12:30


History, 13.10.2019 12:30

SAT, 13.10.2019 12:30

Mathematics, 13.10.2019 12:30

History, 13.10.2019 12:30

Mathematics, 13.10.2019 12:30

History, 13.10.2019 12:30

Spanish, 13.10.2019 12:30