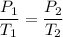Chemistry, 20.11.2020 01:40 nickname0097
Please help with this quiz.
A nitrogen-filled balloon has a pressure of 610 torr at 28°C. What would the pressure
be at 38°C if the volume remained the same?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:30
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 02:50
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3
You know the right answer?
Please help with this quiz.
A nitrogen-filled balloon has a pressure of 610 torr at 28°C. What woul...
Questions








Chemistry, 20.04.2020 22:35

Health, 20.04.2020 22:35

Mathematics, 20.04.2020 22:35


Mathematics, 20.04.2020 22:35

Law, 20.04.2020 22:35

History, 20.04.2020 22:35


Mathematics, 20.04.2020 22:35


Mathematics, 20.04.2020 22:36


History, 20.04.2020 22:36