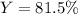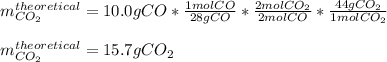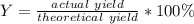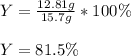Chemistry, 18.11.2020 21:50 mlbaker2381
What is the percent yield for carbon dioxide if 10.0 grams of carbon monoxide yielded
12.81 grams of carbon dioxide from the reaction below?
2 CO + O2 → 2 CO2

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3


Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
What is the percent yield for carbon dioxide if 10.0 grams of carbon monoxide yielded
12.81 grams o...
Questions

Chemistry, 29.01.2021 01:30




English, 29.01.2021 01:30


Social Studies, 29.01.2021 01:30


Social Studies, 29.01.2021 01:30



Mathematics, 29.01.2021 01:30

Health, 29.01.2021 01:30

Mathematics, 29.01.2021 01:30



Mathematics, 29.01.2021 01:30

Mathematics, 29.01.2021 01:30