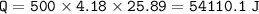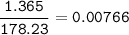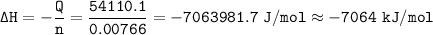When 1.365 g of anthracene, C14H10, is combusted in a bomb calorimeter that has a water jacket containing 500.0 g of water, the temperature of the water increases by 25.89°C. Assuming that the specific heat of water is 4.18 J/(g ∙°C), and that the heat absorption by the calorimeter is negligible, estimate the enthalpy of combustion per mole of anthracene.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
What problem would a person have if the nucleic acid in one of his or her cells were damaged?
Answers: 2


Chemistry, 22.06.2019 14:30
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1
You know the right answer?
When 1.365 g of anthracene, C14H10, is combusted in a bomb calorimeter that has a water jacket conta...
Questions

Mathematics, 12.08.2020 07:01


Mathematics, 12.08.2020 07:01



English, 12.08.2020 07:01





Social Studies, 12.08.2020 07:01




English, 12.08.2020 07:01

Computers and Technology, 12.08.2020 07:01


Social Studies, 12.08.2020 07:01

English, 12.08.2020 07:01

English, 12.08.2020 07:01