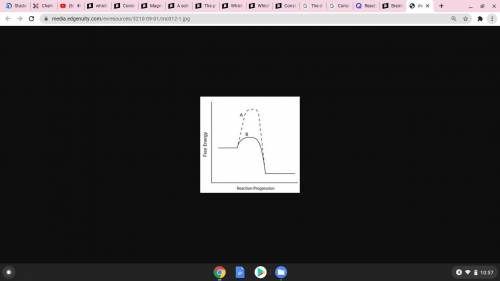
Consider the energy diagram below.
Which line indicates a higher reaction rate?
A because it...


Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 23.06.2019 06:40
How many joules of heat are required to raise thetemperature of 750 g of water from 11.0 °c to 19.0 °c?
Answers: 1

Chemistry, 23.06.2019 09:00
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
You know the right answer?
Questions



Mathematics, 11.06.2020 12:57



Physics, 11.06.2020 12:57


Arts, 11.06.2020 12:57

Mathematics, 11.06.2020 12:57

Biology, 11.06.2020 12:57


Chemistry, 11.06.2020 12:57



Mathematics, 11.06.2020 12:57

Computers and Technology, 11.06.2020 12:57


Computers and Technology, 11.06.2020 12:57


Mathematics, 11.06.2020 12:57