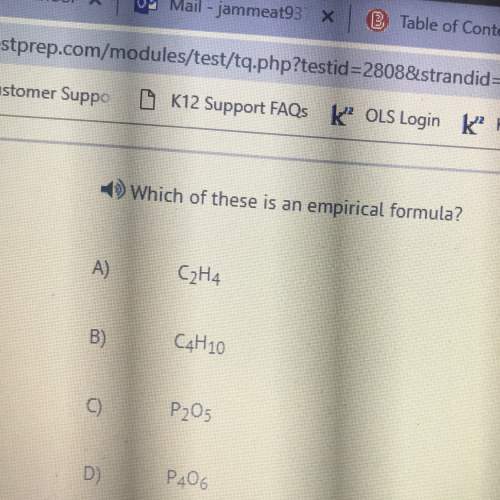
Chemistry, 17.11.2020 18:20 hernandezramirp2pjmh
Please help me ASAP 1.What was your hypothesis?
2.Plot your data as drops of ascorbic acid vs. time.
3.As the concentration of ascorbic acid was increased, did the rate of the formation of the iodine-starch complex increase or decrease? Explain your answer in terms of the chemical reactions involved.
4.Was your hypothesis correct?
5.Make a general rule about the effects of concentration of reactants on reaction rates.
6.For practice, the molecular formula for ascorbic acid is C6H8O6, and you used 6 g in this experiment, calculate the molarity of the ascorbic acid. Now calculate the concentration in moles per drop (assume 1 mL = 20 drops).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1

Chemistry, 21.06.2019 19:00
Asmall amount of a solid is added to water. the observation made after fifteen minutes is shown in the figure. which of these solids has been probably added to water? a) oil b) sand c) sugar d) wood chips
Answers: 1


Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
Please help me ASAP 1.What was your hypothesis?
2.Plot your data as drops of ascorbic acid vs. time...
Questions



Mathematics, 11.04.2021 04:10







History, 11.04.2021 04:10

Mathematics, 11.04.2021 04:10


Mathematics, 11.04.2021 04:10

Mathematics, 11.04.2021 04:10

Mathematics, 11.04.2021 04:10


Mathematics, 11.04.2021 04:10

Social Studies, 11.04.2021 04:10


Chemistry, 11.04.2021 04:20