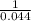19. The reaction 2NOBr (g) → 2 NO (g) + Br2 (g) is a second-order reaction with a rate constant of 0.80 M-1s-1 at 11 °C. If the initial concentration of NOBr is 0.0440 M, the concentration of NOBr after 6.0 seconds is . A) 0.0276 M B) 0.0324 M C) 0.0363 M D) 0.0348 M E) 0.0402 M

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 22.06.2019 04:30
Turbo the snail moves across the ground at a pace of 12 feet per day. if the garden is 48 feet away, how many days will it take for the snail to get there?
Answers: 2

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
19. The reaction 2NOBr (g) → 2 NO (g) + Br2 (g) is a second-order reaction with a rate constant of 0...
Questions



Mathematics, 31.05.2020 23:00


Mathematics, 31.05.2020 23:00

Mathematics, 31.05.2020 23:00





Law, 31.05.2020 23:00

Mathematics, 31.05.2020 23:00





English, 31.05.2020 23:00


Mathematics, 31.05.2020 23:00

Mathematics, 31.05.2020 23:00

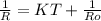
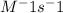
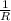 = 0.8*6+
= 0.8*6+