
Chemistry, 17.11.2020 05:40 justaguy15
2.83 kJ are added to a sample of water at 100°C and it completely vaporizes. What is the
mass of the water?
general formula

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 22.06.2019 19:30
Awoman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). what are these specifications in units of centimeters and grams?
Answers: 2
You know the right answer?
2.83 kJ are added to a sample of water at 100°C and it completely vaporizes. What is the
mass of th...
Questions



History, 21.12.2019 01:31

Computers and Technology, 21.12.2019 01:31

Social Studies, 21.12.2019 01:31



History, 21.12.2019 01:31

English, 21.12.2019 01:31


Chemistry, 21.12.2019 01:31



History, 21.12.2019 01:31

Social Studies, 21.12.2019 01:31


Mathematics, 21.12.2019 01:31


English, 21.12.2019 01:31

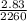 = 0.0013kg
= 0.0013kg

