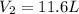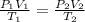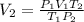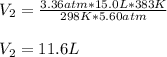Chemistry, 16.11.2020 16:40 shortyyashaun
15.0 L of an ideal gas at 298 K and 3.36 atm are heated to 383 K with a new pressure of 5.60 atm. What is the new volume in liters

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

You know the right answer?
15.0 L of an ideal gas at 298 K and 3.36 atm are heated to 383 K with a new pressure of 5.60 atm. Wh...
Questions


Health, 07.07.2019 05:00

History, 07.07.2019 05:00

History, 07.07.2019 05:00

Mathematics, 07.07.2019 05:00


Physics, 07.07.2019 05:00

Social Studies, 07.07.2019 05:00




Mathematics, 07.07.2019 05:00

History, 07.07.2019 05:00


History, 07.07.2019 05:00

History, 07.07.2019 05:00

History, 07.07.2019 05:00



English, 07.07.2019 05:00