Chemistry, 16.11.2020 05:00 cheesecake1919
10.
Ar(9) deviates more from ideal behavior at extremely high pressures than Ne(g) does. Which of
the following is one reason for this difference?
А A
The particle volume of Ar is greater than that of Ne.
Ar atoms have more valence electrons than Ne atoms have, so Ar atoms have greater interparticle
forces.
The intermolecular forces between Ne atoms are greater than those between Ar atoms.
D Ar atoms are more attracted to the walls of the container than Ne atoms are.
11.
At constant temperature, the behavior of a sample of a real gas more closely approximates that of
an ideal gas as its volume is increased because the

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 12:50
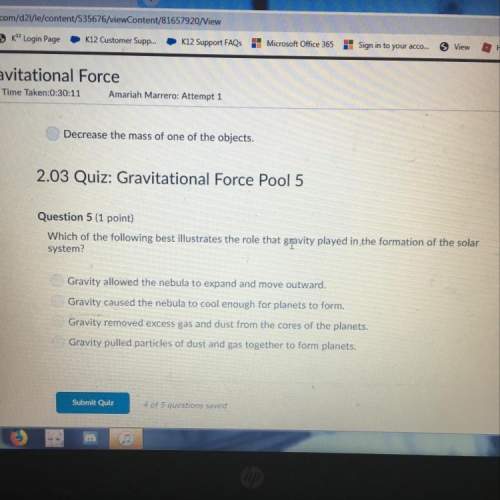
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
10.
Ar(9) deviates more from ideal behavior at extremely high pressures than Ne(g) does. Which of
Questions



Computers and Technology, 25.11.2021 06:40








Chemistry, 25.11.2021 06:40

Computers and Technology, 25.11.2021 06:40

Computers and Technology, 25.11.2021 06:40